Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
Advertisement
Translational Psychiatry volume 15, Article number: 10 (2025)
1
Metrics details
Anxiety disorder, a prevalent mental health issue, is one of the leading causes of disability worldwide. Damage to the blood–brain barrier (BBB) is implicated in anxiety, but its regulatory mechanisms remain unclear. Herein, we show that adrenomedullin 2 (ADM2), a novel angiogenic growth factor, alleviates autistic and anxiety-like behaviors in mice. Based on transcriptome analysis and biochemical analyses, we found that ADM2 facilitates the expression of insulin-like growth factor 2 (IGF-II), which then triggers the activation of the AKT-GSK3β-mTOR signaling pathway via the IGF-II receptor (IGF-IIR), rather than the IGF-I receptor (IGF-IR). Furthermore, as evidenced by increased Evans blue staining and decreased VE-cadherin levels, the BBB exhibited dysfunction in ADM2 knockout mice with anxiety-like behaviors. In in vitro studies, ADM2 administration promoted the expression of VE-cadherin and decreased IGF-II leakage through the endothelial barrier in a BBB model. Taken together, ADM2 may alleviate anxiety-like behavior and social deficits by enhancing BBB integrity and increasing IGF-II levels in the brain. These findings highlight the potential of ADM2 as a therapeutic target for anxiety and related mental disorders.
Anxiety disorder is a prevalent mental health condition worldwide, affecting approximately 25% of the global population. It is characterized by heightened and enduring sensations of fear and apprehension that often exceed the actual threat level, leading to impaired daily functioning [1]. Despite its recurrent nature, only 30% of patients achieve complete remission, highlighting the urgent need to unravel the underlying pathways and signaling molecules involved in emotional dysregulation. Recent research reveals relationship between the blood–brain barrier (BBB) and anxiety-related processes [2]. The BBB is formed by endothelial cells, which seal off the brain from potential harmful signals through tight junctions. Additionally, targeted disruption of the BBB has been found to induce anxiety- and depression-like behaviors [2]. With these findings, a direct association between neurovascular integrity and anxiety susceptibility exists, but the underlying regulatory mechanisms of this association remain unclear.
Adrenomedullin 2 (ADM2; also named intermedin) belongs to the calcitonin/calcitonin gene-related peptide superfamily and exhibits broad expression across the body. Discovered by Roh and Takei in 2004, ADM2 is secreted and exhibits high expression levels specifically within the intermediate pituitary lobe [3, 4]. ADM2 exerts diverse biological effects, including antioxidative stress actions, regulation of hormone secretion, positive inotropic effects, and antiapoptotic properties [5]. Recent evidence suggests that ADM2, acting as a novel angiogenic factor, exhibits a crucial role in modulating angiogenesis. According to Zhang et al, ADM2 promotes vessel remodeling and tumor angiogenesis within of both in vivo and in vitro angiogenesis models [6]. Exogenous ADM2 peptides reactivate quiescent endothelial cells, stimulating proliferation and enhancing blood perfusion [7]. Additionally, ADM2 may contribute to maintaining vascular homeostasis through the upregulation of L-Arg/NOS/NO signaling channel [8]. Furthermore, researches have also shown ADM2 enhances endothelial migration and tube formation through elevating ERK and AKT phosphorylation, along with enhancing vascular endothelial growth factor (VEGF) as well as its receptor synthesis [9]. Furthermore, ADM2 has been found to mitigate organ injuries resulting from vascular hyperpermeability and inflammation [10]. Given this evidence, we hypothesize whether ADM2 might influence anxiety-like behaviors.
In this study, we observed that ADM2 knockout significantly increased the risk of anxiety-like behaviors in mice. Additionally, our research indicated that ADM2 may enhance the expression of VE-cadherin, an endothelial cell adhesion molecule. This effect could potentially decrease the leakage of insulin-like growth factor 2 (IGF-II) from the brain, ultimately improving autistic-like and anxiety-like behaviors in mice. Thus, our study may provide novel insights into the regulatory mechanisms of amygdala ADM2 under anxiety-like behaviors.
Male C57BL/6J mice (wild-type, WT), 8–12 weeks old, selected from Vital River Laboratory Animal Technology Co. Ltd (China). The ADM2 knockout (ADM2-KO) mice were derived using CRISPR/Cas9 technology on a C57BL/6J background [10]. Male and female homozygous mice were multiplied to produce ADM2-KO homozygous offspring. For this study, male homozygous ADM2-KO mice, aged 8–12 weeks, were utilized. All mice were accommodated in a standard facility under 12-h light/12-h dark cycle at a constant temperature with free availability of food and water. The principles in the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals were observed for all experimental programs (NIH publication no. 86–23, revised 1987). Additionally, the experimental protocol of this study was approved by the Institutional Animal Care and Use Committee of the West China Hospital, Sichuan University (Permission Number 2021426A).
The apparatus featured an elevated circular platform with two opposing enclosed quadrants and two open quadrants. Mice were initially introduced to the walled area, and software (ANYmaze, USA) automatically recorded the total distance traveled by the mice within 5 min, as well as the time they spent within open and closed arms.
The overhead plus labyrinth is made of two open arms and two closed arms. Mice were introduced into the central area, and the software (ANYmaze, USA) automatically recorded the total distance traveled by the mice over a 5-min period and the moment held in open and closed arms.
The apparatus was separated into two equal-sized chambers, one brightly lit and the other dark. Allow the mice to free-move among two chambers for a duration of 5 min. Use the software to record the total time taken within each chamber (ANYmaze, USA).
Mice were initially introduced into a 48 × 48 cm arena for a period of 10-min. The arena included a vacant wire-mesh enclosure (10 cm diameter and 15 cm height) positioned in the center. After 2 h, a new mouse was introduced into the wire enclosure and the mice were then placed back into the arena to be observed for an additional 10 min. The time mice spent in the designated social interaction zone (demarcated as a 10 × 10 cm area surrounding the wire-mesh fence) in the presence or absence of the new mouse was recorded using EthoVision XT software (EthoVision, USA).
The first day, mice spent 10 min in the chamber without any objects to acclimate them to the surroundings. The next day, mice were positioned within the same test chamber for another 10 min, on this occasion having two objects alike. Then, the third day, a new object of a different size and color was substituted for either of the familiar objects. The mice were given 10 min to investigate the novel object. Utilizing the EthoVision XT software (EthoVision, USA), the duration spent exploring each object was documented. The exploratory preference was calculated using the formula: time (novel) / (time (novel) + time (familiar)).
During the first day, mice were placed in the chamber for 10 min without any objects. The following day, they were positioned inside the chamber for 10 min along with two completely identical objects. Then, on day three, one of the acquainted objects moved to a diagonal location, maintaining an identical appearance to the original. Afterwards, they were returned to the chamber and given 10 min to investigate the object. EthoVision XT software (EthoVision, USA) recorded the duration spent in exploring both objects. The exploratory preference was calculated using the formula: time (novel)/(time (novel) + time (familiar)).
TMT was performed in a T-shaped maze that contained a start arm as well as two target arms. On day one, mice were given 5 min in which to acclimatize to the maze. On day two, they coached the mice to make movements towards the right arm (old arm) from the starting arm, and to avoid moving to the left arm (new arm). Then, the mice were permitted to randomly probe both arms on third day at will, and the duration stayed on each arm was recorded using software (ANYmaze, USA) during a 5-min test period.
Mice were positioned in the conditioning chamber for a total of 300 s during the training period. Footshocks (2 s, 0.75 mA) were administered at 120, 180, and 240 s. The following morning, or after 7 days, mice were returned to the chamber and allowed to investigate for 180 s. Expression of situational fear memory was assessed through manual recording of frozen acts, which arose from a lack of movement over at least 2 s.
The testing apparatus comprises a 48 × 48 cm open arena with walls of 50-cm high. Every mouse was placed individually in the center of the chamber in order to explore liberally over a period of 5 min. EthoVision XT software (EthoVision, USA) automatically recorded a total distance traveled by mice, and the duration they stayed in the center of the chamber.
Mice were placed inside a 50-cm-high, 20-cm-diameter glass tube containing water and stored at a constant temperature of 25 ± 1 °C for 5 min. During the test period, the immobility time of each mouse was automatically recorded by EthoVision XT software (EthoVision, USA).
Mice were hung 30 cm from the bench and taped about 1 cm from end of their tails. The software (ANYmaze, USA) automatically recorded each mouse’s resting time within the 5-min test.
Mice were anesthetized with sodium pentobarbital at a dosage of 60 mg/kg and fixed on a standard stereotaxic apparatus (RWD Life Science). An incision of the scalp was made to reveal the skull, and bilateral guiding cannulas (RWD Life Science) were permanently embedded in the amygdala (AP, −1.2 mm; ML, ±3.2 mm; DV, −5 mm) using stereotaxic instruments. Following a week of recovery, drug injections were administered at 0.5 μL/min utilizing an infusion pump. After injection, the needle remained for 1 min to ensure that the drug was completely dispersed.
ADM2 (ADM240, which refers to ADM28–47, the mature form of the ADM2 peptide consisting of 40 amino acid residues) and ADM2inh (ADM2 inhibitor, specifically ADM217–47, a truncated form of ADM240 comprising 31 amino acids and acting as a competitive inhibitor of ADM2) were procured from Shinegene (Shanghai, China). Both ADM2 and ADM2inh were dissolved in distilled water (ddH2O) and administered via bilateral injections into the amygdala at a dosage of 0.5 mg/kg per side, following established protocols of prior research [3, 7, 10].
Recombinant IGF-II (792-MG-050, R&D) was solubilized in 0.1% bovine serum albumin (BSA) in PBS. The recombinant IGF-II was administered via bilateral injections into the amygdala at a concentration of 250 ng/μL per side, following established protocols from previous studies [11]. The IGF-I receptor (IGF-IR) antagonist JB1 (J3705, Sigma Aldrich Corp.) was resolved in PBS and administered at an injection concentration of 20 ng/μL, which has been demonstrated to effectively block IGF-IR activity in the amygdala [11]. Anti-IGF-IIR (AF792, R&D) was dissolved in PBS and injected at a concentration of 5 ng/μL, which has been shown to effectively block IGF-IIR activity in the amygdala [11].
Evans blue dye (E2129, Sigma Aldrich) was dissolved in PBS (2% w/v) and administered intraperitoneally to 8-week-old mice at a dosage of 100 mL/25 g body weight. After 2 h post-injection, mice were euthanized, and the brains were harvested as well as inspected for bluish staining. Then, it was incubated for 48 h at 4% paraformaldehyde for extraction of Evans blue dye, with absorption measured at 600 nm [12].
After being anesthetized by sodium pentobarbital, the mice (8 weeks old) were secured in the stereotaxic apparatus. For injecting the virus into the amygdala, tiny cranial holes were created using a skull rotor. AAV2/9-VE-Cadherin-ADM2-mNeonGreen-Flag (1 μL, 0.1 μL/min) was injected bilaterally in amygdala (AP, −1.2 mm; ML, ±3.2 mm; DV, −5 mm). To facilitate virus dispersion, the syringe was left in place for an additional 5 min after each injection before being slowly removed. Following three weeks of postsurgical recovery, the mice underwent subsequent behavioral testing. The accuracy of AAV injections was confirmed in coronal sections (10 μm) using DAPI staining, with images taken with fluorescence microscopy. Statistical analysis excluded mice with improperly positioned injections.
We collected amygdala tissues from both WT and ADM2-KO mice and performed RNA-seq sequencing. Initially, total RNA was estimated by extraction with TRIzol (Invitrogen) and then assessed for RNA integrity using TapeStation (Agilent Technologies, Santa Clara, CA). Subsequently, an NEBNext Ultra Directional RNA Library Prep Kit (New England BioLabs, Ipswich, MA) was employed to synthesize cDNA. Next-generation sequencing was performed on the HiSeq2500 platform (Illumina, San Diego, CA). Statistical package EdgeR was utilized to perform differential gene expression analysis.
Extraction of total RNA was performed by applying TRIzol (Invitrogen), followed by reverse transcription to synthesize cDNA. Subsequently, fluorescence quantitative PCR was analyzed with iTaq™ Universal SYBR® Green One-Step Kit (Bio-Rad, Cat# 1725150). The 2−ΔΔCt method was employed, with the relative level of the target gene normalized to β-Actin. Below are the primers utilized: IGF-II: Forward: 5′-GTTGGTGCTTCTCATCTCTTTG-3′, Reverse: 5′-AAACTGAAGCGTGTCAACAAG-3′; β-actin: Forward: 5′-TCCACGAAACTACCTTCAACTC-3′, Reverse: 5′-CAGTGATCTCCTTCTGCATCC-3′.
Proteins were extracted using RIPA lysis buffer (Thermo Fisher, Cat#89901) coated in protease inhibitors (Thermo Fisher, Cat#A32963). After quantification and denaturation, SDS-PAGE electrophoresis was performed. Subsequently, it was transferred the isolated proteins onto a PVDF membrane (Millipore). Upon blocking by 5% skimmed milk, membranes were incubated overnight at 4 °C to incubate with the primary antibody, and then treated with the secondary antibody labeled with horseradish peroxidase. Immune reaction signals were finally detected using an ECL kit (Millipore) on a chemiluminescence imaging system (CLINX, Shanghai, China). Antibodies were utilized as below: anti-IGF-II (1:1000; Abclonal A2086), anti-IGF-IR (1:1000; Abcam ab182408), anti-IGF-IIR (1:1000; Novus NBP3-16270); anti-p-AKT (1:1000; CST 4060), anti-AKT (1:1000; CST 4685), anti-p-GSK3β (1:1000; CST 5558), anti-GSK3β (1:1000; CST 12456), anti-p-mTOR (1:1000; CST 2974), anti-mTOR (1:1000; CST 2983), anti-p-STAT1 (1:1000; CST 9167), anti-STAT1 (1:1000; CST 9172), anti-GAPDH (1:2000; Abcam ab181602), anti-VE-Cadherin (1:1000; Abcam ab33168).
The mouse brain microvascular endothelial cell line (Bend. 3) was provided by the Shanghai Cell Bank, Chinese Academy of Sciences (iCell-m009). Cultures were grown in Dulbecco’s Modified Eagle’s Medium (DMEM) with 10% fetal bovine serum (Gibco) and 1% penicillin-streptomycin (Gibco) at 37 °C, 5% CO2.
Cell slides were arranged in a 6-well plate, and approximately 5 × 105 Bend. 3 cells were evenly seeded onto the slides and cultured for 24 h. Following this, the cells were fixed with pre-chilled acetone for 20 min, followed by overnight incubation with anti-VE-cadherin antibody (Abcam, Cat# ab205336) at 4 °C. Subsequent steps involved incubating the cells with Alexa Fluor 568-conjugated secondary antibody (Thermo Fisher), followed by staining with DAPI (10 mg/mL) for 10 min. Finally, mounting medium was applied to the slides, and confocal microscopy was used to capture images, followed by quantitative analysis of fluorescence intensity using FIloQuant software.
As previously described [13], the in vitro BBB model was created by sowing a monolayer of Bend.3 cells (1 × 105) into the luminal side of the Transwell instrument and incubating for 48 h. Then, recombinant IGF-II (0.2 μg/μL) was added with ADM2 (2 μM) or ADM2inh (2 μM) into the upper chamber. After 24 h, incubation fluid was gathered in the lower chamber for the assessment of IGF-II levels using an ELISA kit.
All samples were analyzed in a randomized manner. No statistical methods were used for the sample size. No animal was excluded from the analysis. Experimenters were blind to drug treatments. Data were all characterized by GraphPad Prism 7 and depicted as means ± SEMs. Data normality was assessed using Shapiro–Wilk test. Unpaired parametric two-tailed t-tests were applied for comparisons between the two groups. For multiple comparisons, one-way ANOVA analysis and Tukey’s test were used. All findings were statistically significant at p < 0.05 as defined. Figure legends describe the type of statistical test and the definition of significance for various p-values.
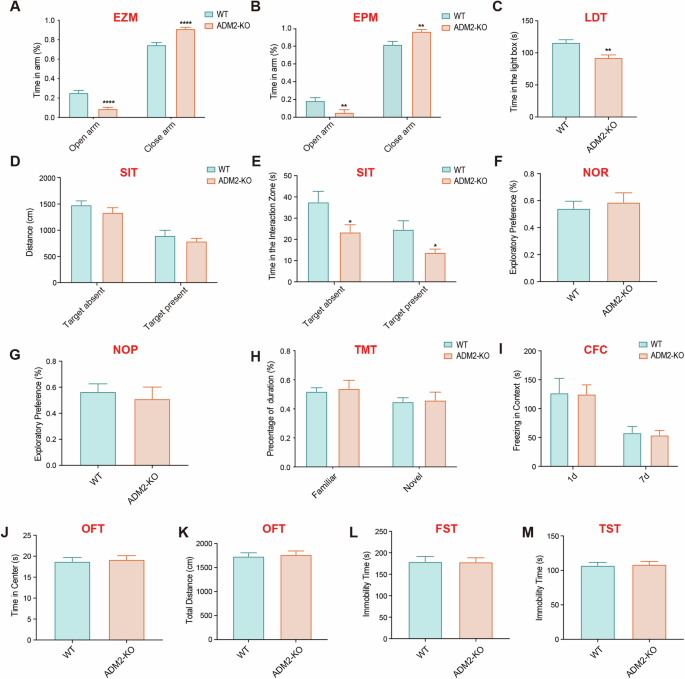
We initially employed the elevated zero maze (EZM) test and the elevated plus maze (EPM) test to evaluate anxiety-like behavior in both WT and ADM2-KO mice. Notably, the time spent in the open arms was significantly reduced in ADM2-KO mice, suggesting an anxiety-like emotional state in these animals (Fig. 1A, B). To further validate this, we conducted the light–dark transition (LDT) test, where ADM2-KO mice exhibited significantly reduced time in the light-box (Fig. 1C). Collectively, these data indicate that ADM2-KO mice may indeed express anxiety-like behavior compared with WT mice.
A The percentage of time spent in the open arm in elevated zero maze (EZM) test (n = 8 per group). B The percentage of time spent in the open arm in elevated plus maze (EPM) test (n = 8 per group). C The amount of time spent in the light box in light dark transition (LDT) test (n = 8 per group). D The distance of mice traveled in the chamber whether the target mice present or absent in social interaction test (SIT) (n = 8 per group). E The amount of time spent in the interaction area whether the target mice present or not in SIT test (n = 8 per group). F The percentage of time spent in exploring new objects in novel object recognition (NOR) test (n = 8 per group). G The percentage of time spent in exploring new location in novel object position (NOP) test (n = 8 per group). H The percentage of time spent in new and old arms during the T maze test (TMT) (n = 8 per group). I The amount of freezing time staying in the context in contextual fear conditioning (CFC) test (n = 8 per group). J The amount of time spent in the center area in the open field test (OFT) (n = 8 per group). K The distance of mice traveled in the chamber in the open field test (OFT) (n = 8 per group). L The amount of immobility time spent in the forced swimming test (FST) (n = 8 per group). M The amount of immobility time spent in the tail suspension test (TST) (n = 8 per group). All data are presented as mean ± SEM. Asterisk indicates the significant difference, which was determined by unpaired parametric two-tailed t-tests. *p < 0.05, **p < 0.01 and ****p < 0.0001.
Given that anxiety-like emotions can potentially lead to deficits in social behavior [14], we proceeded to compare social behaviors between WT and ADM2-KO mice using the social interaction test (SIT). Interestingly, there was no difference in the distance moved between the two groups (Fig. 1D). However, the retention time in the interaction zone was significantly reduced in ADM2-KO mice (Fig. 1E). Furthermore, when exposed to unfamiliar mice, ADM2-KO mice also spent significantly less time in the interaction zone, suggesting decreased social interaction (Fig. 1E).
Next, we assessed memory abilities using the novel object recognition (NOR) test and the novel object position (NOP) test. In the NOR test, there was no significant difference in exploring new objects between the two groups (Fig. 1F). Similarly, ADM2-KO mice did not exhibit spatial memory deficits in the NOP test (Fig. 1G).
To evaluate spatial working memory, we employed the T-maze test (TMT), a classic behavioral paradigm. Surprisingly, there was no significant difference in the time spent in familiar or novel arms between the two groups (Fig. 1H). Additionally, in the contextual fear conditioning (CFC) test conducted on the 1st and 7th days after training, freezing time did not significantly differ between WT and ADM2-KO mice, suggesting that spatial learning and memory abilities were not impaired in ADM2-KO mice (Fig. 1I).
Finally, we investigated depression-like behaviors using the open field test (OFT), forced swimming test (FST), and tail suspension test (TST). In the OFT, there was no significant differences between the two groups of mice in the time they spent in the center or the total distance traveled (Fig. 1J, K). Similarly, the immobility time spent in both the FST and TST did not significantly differ between WT and ADM2-KO mice (Fig. 1L, M), indicating that ADM2 knockout did not induce depression-like behaviors in mice.
Accumulated studies suggest that the amygdala plays a critical role in anxiety-like behaviors [15, 16]. To explore the underlying mechanisms of anxiety-like behavior, we performed RNA sequencing on the amygdala of WT and ADM2-KO mice. We found that the expressions of 383 genes were significantly altered (p < 0.05 and |log2 Fold change|>1) in the amygdala of ADM2-KO mice compared to WT mice, with 357 genes downregulated and 26 genes upregulated (Fig. 2A). The heatmap and box plot analysis indicated a notable decrease in insulin-like growth factor II (IGF-II) expression in ADM2-KO mice (Fig. 2B, C). IGF-II has been reported to play an important role in anxiety-like behaviors [17, 18]. Through RT-PCR and western blot experiments, we confirmed that the mRNA and protein levels of IGF-II were significantly decreased in the amygdala of ADM2-KO mice compared to WT mice (Fig. 2D, E), consistent with the RNA sequencing results.
A The volcano plot showed significant differences in gene expression profiles within the amygdala between WT and ADM2-KO mice (n = 3 per group). B, C The heatmap and box plot indicated a notable decrease in IGF-II expression in AMD2-KO mice. D The mRNA levels of IGF-II gene in the amygdala were quantified by RT-PCR assay (n = 3 per group). E The protein levels of IGF-II, IGF-IR and IGF-IIR in amygdala were assessed by western blot analysis (n = 6 per group). F The protein levels of p-AKT, AKT, p-GSK3β, GSK3β, p-mTOR, mTOR, p-STAT1 and STAT1 in amygdala were assessed by western blot analysis (n = 6 per group). All data are presented as mean ± SEM. Asterisk indicates the significant difference, which was determined by unpaired parametric two-tailed t-tests. *p < 0.05 and **p < 0.01.
IGF-II performs its functions by interacting with specific receptors in the brain, such as IGF-I receptors (IGF-IR) and IGF-II receptors (IGF-IIR, also known as mannose 6-phosphate receptor). As reported, IGF-IR is primarily responsible for mediating the effects of IGF-II on cellular processes such as proliferation, survival, differentiation, and migration. In contrast, IGF-IIR plays a key role in endocytosis, lysosomal degradation, and signal transduction [19,20,21]. Additionally, IGF-IIR has been shown to contribute to the neuroprotective effects of IGF-II [20]. However, western blot analysis showed no significant differences in the levels of IGF-IR or IGF-IIR between ADM2-KO and WT mice (Fig. 2E). We further investigated the downstream AKT-mTOR signaling pathway, which is activated upon IGF-II stimulation and associated with anxiety-like behaviors [22, 23]. Western blot analysis revealed that the expressions of phosphorylated AKT (p-AKT), phosphorylated GSK-3β (p-GSK-3β) and phosphorylated mTOR (p-mTOR) were significantly decreased in ADM2-KO mice, while the levels of AKT, GSK-3β, mTOR, STAT1, and phosphorylated STAT1 (p-STAT1) were unchanged (Fig. 2F).
To further examine the effects of ADM2 and IGF-II, we administered them to the amygdala of ADM2-KO mice and conducted behavioral tests (Fig. 3A). Notably, supplementation with either ADM2 or IGF-II significantly improved anxiety-like behaviors in ADM2-KO mice. This was evidenced by an increased time spent in the open arm in the EZM test (Fig. 3B) and the EPM test (Fig. 3C), as well as more time spent in the light box during the LDT test (Fig. 3D). Additionally, after ADM2 or IGF-II injection, the time spent in the social area significantly increased in the SIT, both with and without the presence of novel mice (Fig. 3E, F). We further examined the levels of IGF-II and its receptors in amygdala of mice. Western blot analysis revealed that the protein level of IGF-II was significantly elevated after either ADM2 or IGF-II injection, while both IGF-IR and IGF-IIR levels remained unchanged (Fig. 3G). Furthermore, upon either ADM2 or IGF-II injection, the protein levels of p-AKT, p-GSK-3β, and p-mTOR were significantly upregulated (Fig. 3H). These results indicate that ADM2 may alleviate anxiety-like behaviors by activating the AKT-mTOR signaling pathway, probably through the upregulation of IGF-II.
A Schematic diagram depicting the timeline of ADM2 or IGF-II administration and subsequent behavioral tests in ADM2-KO mice. B, C The percentage of time spent in the open arm during EZM test and EPM test (n = 8 per group). D The amount of time spent in the light box during the LDT test (n = 8 per group). E, F The distance traveled of mice in the chamber and the amount of time spent in the interaction area during the SIT test (n = 8 for ADM2-KO + PBS group; n = 9 for ADM2-KO + ADM2 group and n = 9 for ADM2-KO + IGF-II group). G The protein levels of IGF-II, IGF-IR and IGF-IIR in amygdala were assessed by western blot analysis (n = 6 per group). H The protein levels of p-AKT, AKT, p-GSK3β, GSK3β, p-mTOR, mTOR, p-STAT1 and STAT1 in amygdala were assessed by western blot analysis (n = 6 per group). All data are presented as mean ± SEM. Asterisk indicates significant difference, determined by one-way ANOVA and Tukey’s test. *p < 0.05 and **p < 0.01.
We then explored the roles of IGF-IR and IGF-IIR by administering their selective inhibitors to the amygdala of mice and conducting behavioral tests (Supplementary Fig. 1A and Fig. 4A). Notably, JB1, a specific IGF-IR inhibitor, did not block the antianxiety effects of IGF-II (Supplementary Fig. 1B–D). Similarly, JB1 failed to reverse the improvements in social deficits induced by IGF-II (Supplementary Fig. 1E, F). Moreover, JB1 could not suppress the IGF-II-triggered activation of the AKT-mTOR signaling pathway (Supplementary Fig. 1G).
A Schematic diagram depicting the timeline of anti-IGF-IIR antibody and/or IGF-II administration and subsequent behavioral tests in ADM2-KO mice. B, C The percentage of time spent in the open arm during EZM test and EPM test (n = 6 per group). D The amount of time spent in the light box during the LDT test (n = 6 per group). E, F The distance of mice traveled in the chamber and the amount of time spent in the interaction area during the SIT test (n = 6 per group). G The protein levels of p-AKT, AKT, p-GSK3β, GSK3β, p-mTOR and mTOR in amygdala were assessed by western blot analysis (n = 6 per group). All data are presented as mean ± SEM. Asterisk indicates significant difference, determined by one-way ANOVA and Tukey’s test. *p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001.
Next, we employed a functional IGF-IIR antibody in the amygdala of ADM2-KO mice, followed by an IGF-II injection (Fig. 4A). The behavior tests showed that the IGF-IIR antibody significantly blocked the antianxiety effects of IGF-II (Fig. 4B–D). Similarly, IGF-IIR antibody administration also abolished the improvement in social deficits induced by IGF-II in ADM2-KO mice (Fig. 4E, F). Furthermore, western blot analysis showed that the IGF-IIR antibody effectively blocked the AKT-mTOR signaling activation induced by IGF-II treatment (Fig. 4G). These data indicate that IGF-II exerts its beneficial effects on anxiety-like behavior and social deficits via IGF-IIR, triggering AKT-mTOR signaling activation.
Next, we investigated whether ADM2 exerts its anti-anxiety effects through IGF-II/IGF-IIR pathway. We administrated ADM2 alone or in combination with the functional IGF-IIR antibody into the amygdala of ADM2-KO mice (Fig. 5A). Behavioral tests showed that the combination of ADM2 with the IGF-IIR antibody significantly reduced the time spent in the open arm during the EZM and EPM tests (Fig. 5B, C), as well as the time spent in the light box during the LDT test (Fig. 5D). Additionally, ADM2’s ability to increase the time spent in the social area during the SIT was also largely counteracted by the IGF-IIR antibody (Fig. 5F). Furthermore, the activation of the AKT-mTOR signaling pathway, evidenced by elevated phosphorylation of AKT, GSK-3β and mTOR following ADM2 administration, was substantially diminished in the presence of the IGF-IIR antibody (Fig. 5G). These findings suggest that ADM2 primarily activates the AKT-mTOR signaling pathway through the IGF-II/IGF-IIR pathway.
A Schematic diagram depicting the timeline of anti-IGF-IIR antibody and/or ADM2 administration and subsequent behavioral tests in ADM2-KO mice. B, C The percentage of time spent in the open arm during EZM test and EPM test (n = 6 per group). D The amount of time spent in the light box during the LDT test (n = 6 per group). E, F The distance of mice traveled in the chamber and the amount of time spent in the interaction area during the SIT test (n = 6 per group). G The protein levels of p-AKT, AKT, p-GSK3β, GSK3β, p-mTOR and mTOR in amygdala were assessed by western blot analysis (n = 6 per group). All data are presented as mean ± SEM. Asterisk indicates significant difference, determined by one-way ANOVA and Tukey’s test. *p < 0.05, **p < 0.01.
ADM2 plays an essential role in several physiological processes, such as regulating blood pressure, promoting vasodilation, and modulating angiogenesis [5]. Building on this, we sought to determine whether ADM2 influence the function of the blood–brain barrier (BBB). Our results show a marked increase in Evans blue dye in the brains of ADM2-KO mice compared to WT mice (Fig. 6A–C), indicating increased BBB permeability in ADM2-KO mice. Furthermore, the levels of VE-cadherin were significantly decreased in the brains of ADM2-KO mice (Fig. 6D), further suggesting the compromised BBB integrity in ADM2-KO mice. Additionally, in Bend.3 cells, the protein levels of VE-cadherin were significantly upregulated upon stimulation with ADM2 plus IGF-II but downregulated upon stimulation with ADM2inh plus IGF-II, as evidenced by immunofluorescence staining and western blot analysis (Fig. 6E, F). Finally, in an in vitro BBB model (Fig. 6G), ADM2 treatment resulted in a substantial reduction of IGF-II leakage into the lower chamber (Fig. 6H). These findings indicate that ADM2 plays a critical role in maintaining BBB function and decreasing IGF-II leakage from the brain.
A, B Image of mice taken 2 h after Evans Blue injection. C The brains of mice photographed 2 h after Evans Blue injection, and absorbances at 600 nm were detected in each group (n = 6 per group). Data are shown as mean ± SEM. Unpaired parametric two-tailed t-tests were used for statistical analysis. D The protein levels of VE-Cadherin in amygdala were assessed by western blot analysis (n = 6 per group). Data are shown as mean ± SEM. Unpaired parametric two-tailed t-tests were used for statistical analysis. E The levels of VE-Cadherin in Bend.3 cells were assessed by immunofluorescence staining (n = 3 per group). F The protein levels of VE-Cadherin in Bend.3 cells were assessed by western blot analysis (n = 3 per group). G Schematic diagram of the trans-well assay, with Bend.3 cells seeded in the upper compartment. H The levels of IGF-II in the lower compartment were assessed by ELISA. Data are presented as mean ± SEM. Asterisk indicates the significant difference, determined by one-way ANOVA and Tukey’s test. *p < 0.05, ***p < 0.001 and ****p < 0.0001.
Finally, ADM2 was overexpressed in amygdala endothelial cells through AAV2/9-VE-cadherin-ADM2-mNeonGreen injection, as verified by the visualized expression of mNeonGreen (Fig. 7A). Overexpression of ADM2 in the amygdala endothelial cells significantly increased the time spent in the open arms in EZM and EPM tests and the time spent in the light box in the LDT test in ADM2-KO mice (Fig. 7B–D). Additionally, overexpression of ADM2 significantly increased the time spent in the social zone in the SIT test, whether the novel mice were present or not (Fig. 7E, F). Furthermore, the level of IGF-II increased, and the AKT-mTOR signaling pathway was activated in the amygdala of ADM2-KO mice after overexpression of ADM2 in amygdala endothelial cells (Fig. 7G). These results provide in vivo evidence that overexpression of ADM2 in amygdala endothelial cells can alleviate anxiety-like behaviors via increasing IGF-II levels and activating the downstream AKT-mTOR signaling pathway.
A Schematic diagram depicting the timeline of AAV2/9-VE-Cadherin-ADM2-mNeonGreen administration and subsequent behavioral tests in ADM2-KO mice. B, C The percentage of time spent in the open arm during EZM test and EPM test (n = 7 per group). D The amount of time spent in the light box during the LDT test (n = 7 per group). E, F The distance of mice traveled in the chamber and the amount of time spent in the interaction area during the SIT test (n = 6 per group). G The protein levels of IGF-II, p-AKT, AKT, p-GSK3β, GSK3β, p-mTOR and mTOR in amygdala were assessed by western blot analysis (n = 6 per group). All data are presented as mean ± SEM. Asterisk indicates significant difference, determined by unpaired parametric two-tailed t-tests. *p < 0.05, **p < 0.01.
Adrenomedullin 2 (ADM2) plays a pivotal role in maintaining the body’s homeostasis, particularly in various vascular diseases such as atherosclerosis [24], hypertension [25], and vascular calcification [26]. Existing research indicates that ADM2 strengthens cell-cell adhesion and promotes the rearrangement of the VE-cadherin and actin cytoskeletons, thereby exerting a protective effect on endothelial barrier function [27]. The blood–brain barrier (BBB), as one of the most classic and important endothelial barriers, has been reported to be dysfunctional in the pathogenesis of anxiety [28]. However, there have been few studies on the effects of ADM2 on anxiety-related behaviors in mice. Notably, ADM2 knockout induced anxiety-like behaviors and reduced social interaction. Intriguingly, direct injection of ADM2 into the amygdala significantly ameliorated these anxiety-like behaviors, suggesting a protective role for ADM2 in anxiety-related responses. Mechanistically, we observed compromised BBB integrity in ADM2-KO mice, evidenced by increased permeability (as indicated by Evans blue leakage) and downregulated expression of cell-cell junctions (VE-cadherin). However, overexpression of ADM2 in amygdala endothelial cells effectively mitigated these injuries, reinforcing the importance of ADM2 in maintaining BBB stability and integrity.
Our investigation revealed a significant downregulation of IGF-II in the amygdala of ADM2-KO mice, primarily associated with transcriptional regulation and increased leakage due to blood-brain barrier disruption. IGF-II is a pleiotropic polypeptide widely distributed in various tissues, with particularly high expression levels in the brain [21, 29, 30]. It plays a pivotal role in diverse diseases, including cancer, metabolic disorders, and cardiovascular conditions, acting through different receptors [31,32,33]. Specifically, within the central nervous system, IGF-II exerts neuroprotective effects, modulating brain growth, development, and high-order functions [11, 34], including memory consolidation and enhancement [35,36,37]. Interestingly, previous studies have linked IGF-II downregulation to depression. In a mouse model of depression, IGF-II expression was downregulated, and its levels increased as the depressive phenotype improved [18, 38]. In our study, we demonstrated that IGF-II was significantly downregulated in mice exhibiting anxiety-like responses and suggested that supplementation with IGF-II markedly reversed these manifestations, thereby identifying the protective role of IGF-II in anxiety-like behaviors.
Furthermore, we investigated the contribution of IGF receptors to the effects of IGF-II in our study. Although the expression levels of both IGF-IR and IGF-IIR remained relatively stable, the antianxiety effects of IGF-II were primarily blocked by the IGF-IIR antibody, but not by the IGF-IR inhibitor (JB1). This finding underscores the critical role of IGF-IIR in mediating these effects, while IGF-IR appears less relevant in this context. IGF-IIR, a single-transmembrane protein, comprises 15 homologous domains and a diminutive cytoplasmic tail. Abundantly present in neurons, it fulfills crucial functions in endosomal trafficking, protein synthesis, and lysosomal targeting [39, 40]. Notably, IGF-IIR has emerged as a therapeutic target for various CNS disorders, as several pathologies have shown improvement upon treatment with IGF-II or other ligands that activate IGF-IIR [41,42,43].
Additionally, our research unveiled that IGF-II-induced activation of IGF-IIR results in heightened activation of the AKT-GSK-3β-mTOR signaling cascade. This pathway assumes a pivotal role in neuronal cell proliferation, migration, and plasticity. Dysregulation of AKT-mTOR signaling has been implicated in various mental illnesses, including depression [44], Alzheimer’s disease [45], post-traumatic stress disorder (PTSD) [23], and anxiety-like behaviors [46]. Upon activation, AKT undergoes phosphorylation, which subsequently phosphorylates GSK-3β at the critical Ser 9 site, inhibiting its activation. Notably, GSK-3β also contributes significantly to neural health, particularly in the context of neuronal responses to stress and synapse maintenance [45].
In summary, our data demonstrate that ADM2 knockout induces abnormal neurobehaviors in mice, primarily characterized by anxiety-like behaviors and social behavior deficits. Interestingly, these effects do not extend to memory impairments or depression-like behavior. Furthermore, we confirmed that ADM2 knockout leads to downregulation of IGF-II and BBB impairment, resulting in inactivation of the AKT-mTOR signaling pathway, ultimately contributing to anxiety-like behaviors.
The datasets generated during this study are accessible upon request to the corresponding author.
GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the global burden of disease study 2017. Lancet. 2018;392:1789–858.
Article Google Scholar
Dion-Albert L, Cadoret A, Doney E, Kaufmann FN, Dudek KA, Daigle B, et al. Vascular and blood-brain barrier-related changes underlie stress responses and resilience in female mice and depression in human tissue. Nat Commun. 2022;13:164.
Article PubMed PubMed Central Google Scholar
Roh J, Chang CL, Bhalla A, Klein C, Hsu SY. Intermedin is a calcitonin/calcitonin gene-related peptide family peptide acting through the calcitonin receptor-like receptor/receptor activity-modifying protein receptor complexes. J Biol Chem. 2004;279:7264–74.
Article PubMed Google Scholar
Takei Y, Inoue K, Ogoshi M, Kawahara T, Bannai H, Miyano S. Identification of novel adrenomedullin in mammals: a potent cardiovascular and renal regulator. FEBS Lett. 2004;556:53–8.
Article PubMed Google Scholar
Yang Z, Li H, Wu P, Li Q, Yu C, Wang D, et al. Multi-biological functions of intermedin in diseases. Front Physiol. 2023;14:1233073.
Article PubMed PubMed Central Google Scholar
Zhang W, Wang LJ, Xiao F, Wei Y, Ke W, Xin HB. Intermedin: a novel regulator for vascular remodeling and tumor vessel normalization by regulating vascular endothelial-cadherin and extracellular signal-regulated kinase. Arterioscler Thromb Vasc Biol. 2012;32:2721–32.
Article PubMed Google Scholar
Wang LJ, Xiao F, Kong LM, Wang DN, Li HY, Wei YG, et al. Intermedin enlarges the vascular lumen by inducing the quiescent endothelial cell proliferation. Arterioscler Thromb Vasc Biol. 2018;38:398–413.
Article PubMed Google Scholar
Yang JH, Pan CS, Jia YX, Zhang J, Zhao J, Pang YZ, et al. Intermedin1-53 activates L-arginine/nitric oxide synthase/nitric oxide pathway in rat aortas. Biochem Biophys Res Commun. 2006;341:567–72.
Article PubMed Google Scholar
Smith RS Jr., Gao L, Bledsoe G, Chao L, Chao J. Intermedin is a new angiogenic growth factor. Am J Physiol Heart Circ Physiol. 2009;297:H1040–7.
Article PubMed PubMed Central Google Scholar
Xiao F, Wang D, Kong L, Li M, Feng Z, Shuai B, et al. Intermedin protects against sepsis by concurrently re-establishing the endothelial barrier and alleviating inflammatory responses. Nat Commun. 2018;9:2644.
Article PubMed PubMed Central Google Scholar
Chen DY, Stern SA, Garcia-Osta A, Saunier-Rebori B, Pollonini G, Bambah-Mukku D, et al. A critical role for IGF-II in memory consolidation and enhancement. Nature. 2011;469:491–7.
Article PubMed PubMed Central Google Scholar
Recuenco FC, Takano R, Chiba S, Sugi T, Takemae H, Murakoshi F, et al. Lambda-carrageenan treatment exacerbates the severity of cerebral malaria caused by plasmodium berghei ANKA in BALB/c mice. Malar J. 2014;13:487.
Article PubMed PubMed Central Google Scholar
Naik P, Cucullo L. In vitro blood-brain barrier models: current and perspective technologies. J Pharm Sci. 2012;101:1337–54.
Article PubMed Google Scholar
Yoo JY, Kim HB, Lee YJ, Kim YJ, Yoo SY, Choi Y, et al. Neuregulin-1 reverses anxiety-like behavior and social behavior deficits induced by unilateral micro-injection of CoCl(2) into the ventral hippocampus (vHPC). Neurobiol Dis. 2023;177:105982.
Article PubMed Google Scholar
Fan KQ, Li YY, Wang HL, Mao XT, Guo JX, Wang F, et al. Stress-induced metabolic disorder in peripheral CD4+ T cells leads to anxiety-like behavior. Cell. 2019;179:864–79. e19
Article PubMed Google Scholar
Pawlak R, Magarinos AM, Melchor J, McEwen B, Strickland S. Tissue plasminogen activator in the amygdala is critical for stress-induced anxiety-like behavior. Nat Neurosci. 2003;6:168–74.
Article PubMed Google Scholar
Li CQ, Luo YW, Bi FF, Cui TT, Song L, Cao WY, et al. Development of anxiety-like behavior via hippocampal IGF-2 signaling in the offspring of parental morphine exposure: effect of enriched environment. Neuropsychopharmacology. 2014;39:2777–87.
Article PubMed PubMed Central Google Scholar
Li Y, Chen Y, Gao X, Zhang Z. The behavioral deficits and cognitive impairment are correlated with decreased IGF-II and ERK in depressed mice induced by chronic unpredictable stress. Int J Neurosci. 2017;127:1096–103.
Article PubMed Google Scholar
Bondy CA, Lee WH. Patterns of insulin-like growth factor and IGF receptor gene expression in the brain. functional implications. Ann N Y Acad Sci. 1993;692:33–43.
Article PubMed Google Scholar
Wang Y, MacDonald RG, Thinakaran G, Kar S. Insulin-like growth factor-II/Cation-independent mannose 6-phosphate receptor in neurodegenerative diseases. Mol Neurobiol. 2017;54:2636–58.
Article PubMed Google Scholar
Beletskiy A, Chesnokova E, Bal N. Insulin-like growth factor 2 as a possible neuroprotective agent and memory enhancer-Its comparative expression, processing and signaling in mammalian CNS. Int J Mol Sci. 2021;22:1849.
Article PubMed PubMed Central Google Scholar
Wang X, Lin L, Lan B, Wang Y, Du L, Chen X, et al. IGF2R-initiated proton rechanneling dictates an anti-inflammatory property in macrophages. Sci Adv. 2020;6:eabb7389.
Article PubMed PubMed Central Google Scholar
Gao F, Wang J, Yang S, Ji M, Zhu G. Fear extinction induced by activation of PKA ameliorates anxiety-like behavior in PTSD mice. Neuropharmacology. 2023;222:109306.
Article PubMed Google Scholar
Zhang X, Gu L, Chen X, Wang S, Deng X, Liu K, et al. Intermedin ameliorates atherosclerosis in ApoE null mice by modifying lipid profiles. Peptides. 2012;37:189–93.
Article PubMed Google Scholar
Kobayashi Y, Liu YJ, Gonda T, Takei Y. Coronary vasodilatory response to a novel peptide, adrenomedullin 2. Clin Exp Pharmacol Physiol. 2004;31:S49–50. Suppl 2
Article PubMed Google Scholar
Chang JR, Duan XH, Zhang BH, Teng X, Zhou YB, Liu Y, et al. Intermedin1-53 attenuates vascular smooth muscle cell calcification by inhibiting endoplasmic reticulum stress via cyclic adenosine monophosphate/protein kinase a pathway. Exp Biol Med. 2013;238:1136–46.
Article Google Scholar
Aslam M, Pfeil U, Gündüz D, Rafiq A, Kummer W, Piper HM, et al. Intermedin (adrenomedullin2) stabilizes the endothelial barrier and antagonizes thrombin-induced barrier failure in endothelial cell monolayers. Br J Pharmacol. 2012;165:208–22.
Article PubMed PubMed Central Google Scholar
Villalba N, Ma Y, Gahan SA, Joly-Amado A, Spence S, Yang X, et al. Lung infection by pseudomonas aeruginosa induces neuroinflammation and blood-brain barrier dysfunction in mice. J Neuroinflamm. 2023;20:127.
Article Google Scholar
DeChiara TM, Efstratiadis A, Robertson EJ. A growth-deficiency phenotype in heterozygous mice carrying an insulin-like growth factor II gene disrupted by targeting. Nature. 1990;345:78–80.
Article PubMed Google Scholar
Louvi A, Accili D, Efstratiadis A. Growth-promoting interaction of IGF-II with the insulin receptor during mouse embryonic development. Dev Biol. 1997;189:33–48.
Article PubMed Google Scholar
Scalia P, Giordano A, Williams SJ. The IGF-II-Insulin receptor Isoform-A autocrine signal in cancer: actionable perspectives. Cancers. 2020;12:366.
Article PubMed PubMed Central Google Scholar
Livingstone C, Borai A. Insulin-like growth factor-II: its role in metabolic and endocrine disease. Clin Endocrinol. 2014;80:773–81.
Article Google Scholar
Faienza MF, Santoro N, Lauciello R, Calabrò R, Giordani L, Di Salvo G, et al. IGF2 gene variants and risk of hypertension in obese children and adolescents. Pediatr Res. 2010;67:340–4.
Article PubMed Google Scholar
Agis-Balboa RC, Arcos-Diaz D, Wittnam J, Govindarajan N, Blom K, Burkhardt S, et al. A hippocampal insulin-growth factor 2 pathway regulates the extinction of fear memories. Embo J. 2011;30:4071–83.
Article PubMed PubMed Central Google Scholar
Stern SA, Kohtz AS, Pollonini G, Alberini CM. Enhancement of memories by systemic administration of insulin-like growth factor II. Neuropsychopharmacology. 2014;39:2179–90.
Article PubMed PubMed Central Google Scholar
Stern SA, Chen DY, Alberini CM. The effect of insulin and insulin-like growth factors on hippocampus- and amygdala-dependent long-term memory formation. Learn Mem. 2014;21:556–63.
Article PubMed PubMed Central Google Scholar
Steinmetz AB, Johnson SA, Iannitelli DE, Pollonini G, Alberini CM. Insulin-like growth factor 2 rescues aging-related memory loss in rats. Neurobiol Aging. 2016;44:9–21.
Article PubMed PubMed Central Google Scholar
Poggini S, Golia MT, Alboni S, Milior G, Sciarria LP, Viglione A, et al. Combined fluoxetine and metformin treatment potentiates antidepressant efficacy increasing IGF2 expression in the dorsal hippocampus. Neural Plast. 2019;2019:4651031.
Article PubMed PubMed Central Google Scholar
Alberini CM. IGF2 in memory, neurodevelopmental disorders, and neurodegenerative diseases. Trends Neurosci. 2023;46:488–502.
Article PubMed PubMed Central Google Scholar
Ghosh P, Dahms NM, Kornfeld S. Mannose 6-phosphate receptors: new twists in the tale. Nat Rev Mol Cell Biol. 2003;4:202–12.
Article PubMed Google Scholar
Kar S, Poirier J, Guevara J, Dea D, Hawkes C, Robitaille Y, et al. Cellular distribution of insulin-like growth factor-II/mannose-6-phosphate receptor in normal human brain and its alteration in Alzheimer’s disease pathology. Neurobiol Aging. 2006;27:199–210.
Article PubMed Google Scholar
Mackay KB, Loddick SA, Naeve GS, Vana AM, Verge GM, Foster AC. Neuroprotective effects of insulin-like growth factor-binding protein ligand inhibitors in vitro and in vivo. J Cereb Blood Flow Metab. 2003;23:1160–7.
Article PubMed Google Scholar
Martin-Montañez E, Pavia J, Santin LJ, Boraldi F, Estivill-Torrus G, Aguirre JA, et al. Involvement of IGF-II receptors in the antioxidant and neuroprotective effects of IGF-II on adult cortical neuronal cultures. Biochim Biophys Acta. 2014;1842:1041–51.
Article PubMed Google Scholar
Kitagishi Y, Kobayashi M, Kikuta K, Matsuda S. Roles of PI3K/AKT/GSK3/mTOR pathway in cell signaling of mental illnesses. Depress Res Treat. 2012;2012:752563.
PubMed PubMed Central Google Scholar
Gabbouj S, Ryhänen S, Marttinen M, Wittrahm R, Takalo M, Kemppainen S, et al. Altered insulin signaling in Alzheimer’s disease brain—special emphasis on PI3K-Akt pathway. Front Neurosci. 2019;13:629.
Article PubMed PubMed Central Google Scholar
Ostovan VR, Amiri Z, Moezi L, Pirsalami F, Esmaili Z, Moosavi M. The effects of subchronic agmatine on passive avoidance memory, anxiety-like behavior and hippocampal Akt/GSK-3β in mice. Behav Pharmacol. 2022;33:42–50.
Article PubMed Google Scholar
Download references
This work was financially supported by the National Natural Science Foundation of China (82102301) and Natural Science Foundation of Sichuan (2024NSFSC0732).
These authors contributed equally: Denian Wang, Zhi Yang.
Precision Medicine Research Center, Precision Medicine Key Laboratory of Sichuan Province, State Key Laboratory of Respiratory Health and Multimorbidity, Frontiers Science Center for Disease-related Molecular Network, West China Hospital, Sichuan University, Chengdu, Sichuan, China
Denian Wang, Pengfei Wu, Qingyan Li, Ying Yang, Yuefan Du & Mengwei Jiang
Department of Nephrology, West China Hospital, Sichuan University, Chengdu, Sichuan, China
Zhi Yang
Frontiers Science Center for Disease-related Molecular Network, Laboratory of Omics Technology and Bioinformatics. West China Hospital, Sichuan University, Chengdu, Sichuan, China
Chunyan Yu
Department of Neurosurgery, West China Hospital of Sichuan University, Chengdu, Sichuan, China
Junpeng Ma
Department of Neurosurgery, West China Tianfu Hospital of Sichuan University, Chengdu, Sichuan, China
Junpeng Ma
You can also search for this author in PubMed Google Scholar
You can also search for this author in PubMed Google Scholar
You can also search for this author in PubMed Google Scholar
You can also search for this author in PubMed Google Scholar
You can also search for this author in PubMed Google Scholar
You can also search for this author in PubMed Google Scholar
You can also search for this author in PubMed Google Scholar
You can also search for this author in PubMed Google Scholar
You can also search for this author in PubMed Google Scholar
All authors contributed to the study and approved the final manuscript. Denian Wang: Conceptualization, Data curation, Formal analysis, Methodology, Funding acquisition, Writing-Original draft preparation. Zhi Yang: Data curation, Formal analysis, Investigation, Writing-Original draft preparation. Pengfei Wu: Formal analysis, Investigation, Visualization. Qingyan Li: Formal analysis, Investigation. Chunyan Yu: Investigation, Validation. Ying Yang: Data curation, Investigation. Yuefan Du: Data curation, Investigation, Mengwei Jiang: Data curation, Investigation. Junpeng Ma: Conceptualization, Data curation, Funding acquisition, Supervision, Writing-Reviewing and Editing.
Correspondence to Junpeng Ma.
The authors declare no competing interests.
All methods were performed in accordance with the relevant guidelines and regulations. The experimental protocol of this study was approved by the Institutional Animal Care and Use Committee of the West China Hospital, Sichuan University (Permission Number 2021426A).
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
Reprints and permissions
Wang, D., Yang, Z., Wu, P. et al. Adrenomedullin 2 attenuates anxiety-like behaviors by increasing IGF-II in amygdala and re-establishing blood–brain barrier. Transl Psychiatry 15, 10 (2025). https://doi.org/10.1038/s41398-025-03229-2
Download citation
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-025-03229-2
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Advertisement
© 2025 Springer Nature Limited

More Stories
Anatomy of a Scam
Climate and Environmental Sustainability Within the IETF and IRTF
From Commitments to Practice: Internet Society’s Priorities for WSIS+20 Implementation